Diagnosis of Pulmonary Embolism with d-Dimer Adjusted to Clinical Probability
N Engl J Med, 381 (22), 2125-2134
28 November 2019
Clive Kearon, Kerstin de Wit, Sameer Parpia, Sam Schulman, Marc Afilalo, Andrew Hirsch, Frederick A Spencer, Sangita Sharma, Frédérick D’Aragon, Jean-François Deshaies, Gregoire Le Gal, Alejandro Lazo-Langner, Cynthia Wu, Lisa Rudd-Scott, Shannon M Bates, Jim A Julian, PEGeD Study Investigators
PMID: 31774957
DOI: 10.1056/NEJMoa1909159
ClinicalTrials.gov: NCT02483442
Introduction
Venous thromboembolism is a spectrum of clinical diagnoses and contains deep vein thrombosis and pulmonary embolism. These can be symptomatic or asymptomatic and their diagnosis and management depends on clinical presentation, severity (extension) and risk factors. It is a common diagnosis encountered in primary, secondary or tertiary practice.
In United Kingdom current guidelines by NICE recommend two-level Wells’ score for suspected pulmonary embolism. It uses combination of Wells’ score and D-dimer and stratifies patients into two categories – PE likely (≥ 4.5 points) or PE unlikely (0 – 4.0 points). Negative D-dimer can rule out PE when pretest probability is low, however, NICE does not define cut off for negative D-dimer. Common cut off is 500 ng/ml, although some institutions may use age adjusted D-dimer. This practice leads to use of chest imaging in form of CTPA in all positive D-dimers and all cases with Wells’ score ≥ 4.5 points.
Pulmonary Embolism Graduated D-Dimer (PEGeD) Study looked at the incidence of venous thromboembolism using new diagnostic algorithm. Instead of two-level Wells’ score, D-dimer was adjusted to clinical probability (low, moderate or high) using on Wells’ score for pulmonary embolism. In low probability group cut off of D-dimer was raised to 1000 ng/ml and in high risk groups no D-dimer was used. This new approach might decrease the need for chest imaging without compromising safety.
Methods
Study design
Funded by Canadian Institutes of Health Research (CIHR) this study was a prospective, multi-centre, observational study with independent adjudication of outcomes.
The intention of the study was to establish if new diagnostic strategy with D-dimer adjusted to clinical probability can reduce the use of diagnostic imaging for pulmonary embolism without compromising safety. Patients were followed-up to assess the occurrence of venous thromboembolism (proximal DVT or PE).
Did the study addressed a clearly focused issue?
Yes.
Total of 2017 patients from university-based clinical centres in Canada were enrolled prospectively and results evaluated by central adjudication committee. Members of the committee were unaware of the initial results or anticoagulation therapy. Patients provided verbal or written consent within 7 days of undergoing diagnostic testing.
Was the cohort recruited in an acceptable way?
Yes. Patients were enrolled prospectively and were consented. However, it is not clear what was the total number of patients with suspected PE in the involved centres and this might present a selection bias.
Was the exposure adequately measured to minimise bias?
No. It is not clear what was the total number of patients with suspected PE in the involved centres and this might present a selection bias.
Eligibility
Inpatients or outpatients were eligible for study if they had clinically suspected pulmonary embolism. However, it should be noted in the final analysis of 2017 patients, 2016 were outpatients (from emergency department or outpatient clinics) and only one was inpatient.
Patients were excluded if they:
- were younger than 18 years of age;
- had major surgery in the last 3 weeks;
- were anticoagulated for ≥ 24 hours before D-dimer was measured;
- had D-dimer result before Wells score was calculated;
- undergone chest scanning (CTPA or V/Q scan) before Wells score or outside of the protocol (when negative D-dimer) or had CT chest with contrast for another reason.
- had life expectancy of less than 3 months;
- could not be followed up due to geographical reasons;
- were pregnant or did not provide consent.
Study protocol
In all suspected cases clinical pretest probability using Wells’ criteria for pulmonary embolism was assessed and patients categorised as either low (0 – 4.0 points), moderate (4.5 – 6 points) or high probability (6.5 – 12.5 points). Patients with high probability underwent diagnostic CT pulmonary angiogram without D-dimer assay. Patients with low or moderate probability had D-dimer tested. D-dimer was considered negative if less than 1000 ng/ml in low probability group or less than 500 ng/ml in moderate probability group and no further imaging was done. If D-dimer was positive (≥ 1000 ng/ml in low probability group or ≥ 500 ng/ml in moderate probability group) patients underwent chest imaging – either CTPA or ventilation/perfusion scan depending on the discretion of the physician. All patients were followed up at 90 days after initial testing either over the phone or in clinic.
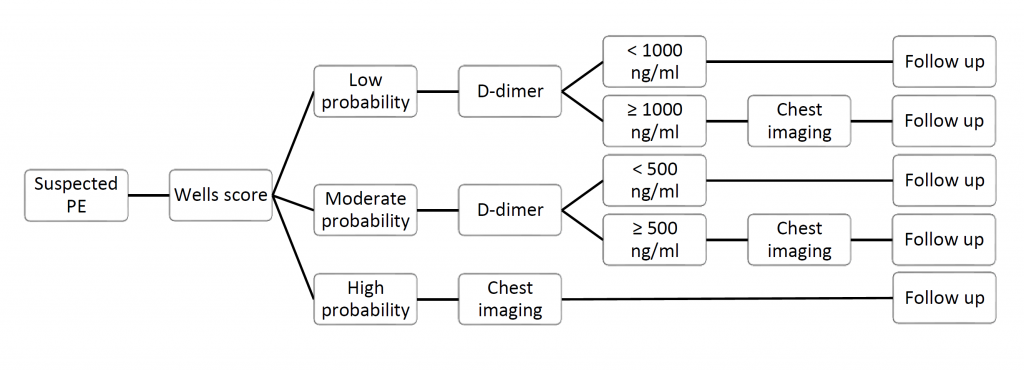
Outcome measures
Primary outcome was confirmed symptomatic venous thromboembolism (proximal DVT or PE). Secondary outcomes were minor and major bleeding, all causes and specific causes of deaths and all other cases of venous thromboembolism that did not satisfy the primary outcome.
Additionally, the percentage of patients who would undergo chest imaging and D-dimer testing with PEGeD strategy was compared with other diagnostic strategies (standard, age-adjusted and YEARS).
Was the outcome adequately measured to minimise bias?
Yes.
Have the authors identified all important confounding factors?
No.
Have they taken account of the confounding factors in the design and/or analysis?
No. For example provoked (i.e. history of cancer) or unprovoked VTE or prior use of anticoagulants was not taken into account.
Analysis
Outcomes were summarised as point estimates and percentages with 95% confidence intervals. The Agresti-Min method was used to obtain 95% CI for paired differences in patients undergoing imaging compared to other strategies.
All outcomes were analysed by central adjudication committee and their members were not aware of the results of initial diagnostic testing or use of anticoagulant therapy.
Follow-up
All enrolled patients were followed-up for a period of 90 days +/- 7 days from the initial diagnostic testing and were assessed from venous thromboembolism.
Was the follow up of subjects complete enough?
Yes. All of the patients were followed-up and assessed for VTE.
Was the follow up of subjects long enough?
Yes. 90 days (3 months) follow-up should be long enough for detection and/or development of VTE.
Results
Population
Mean age was 52 years. 66.2% (1335) were female and mean weight was 81 kg. Median days of symptoms was 5 days (range 0 – 365 days).
Primary outcome
Of 2017 patients in total, 1752 or 86.9% had low pretest probability (Wells’ score 0 – 4.0 points). In this group of patients:
- 1285 patients had negative D-dimer test (< 1000 ng/ml). One of the patients underwent ventilation-perfusion scan of the chest – no PE. Nine of the patients were lost in the follow-up period. All followed-up patients had no occurrence of VTE at 90 days.
- 467 patients had positive D-dimer test (≥ 1000 ng/ml). They underwhen chest imaging and 87 of them had PE. 380 patients had no PE on initial testing. 4 patients were lost in the follow up period (none had PE at initial testing). At follow up in 90 days, 2 patients developed venous thromboembolism – one of the patients developed DVT (there was no PE on initial testing and was not anticoagulated) and one had recurrent PE (there was a PE on initial testing and was anticoagulated).
In 218 patients (10.8%) with moderate pretest probability:
- 40 patients had negative D-dimer test (<500 ng/ml). None of them had VTE at 90 days. None were lost to follow-up.
- 178 patients with positive D-dimer underwent chest imaging and 43 of them had PE on initial testing and 42 were anticoagulated (1 did not receive anticoagulation, reason is not stated). None of the patients were lost in follow-up period and none had VTE at 90 days.
In combined subgroup of 1970 patients with low or moderate pretest probability (97.7% of the cohort), 1325 patients (67.3%) had negative D-dimer and were not anticoagulated – none (0%; 95% CI 0%-0.29%) of them had VTE at 90 days.
47 patients (2.3%) had high pretest probability (Wells’ ≥6.5 points) and underwent chest imaging without awaiting D-dimer. 19 patients (40% of the subgroup) had PE and 28 patients did not. 1 of the patients with PE did not receive anticoagulation due to thrombocytopenia. All of the patients were followed-up at 90 days and none had VTE.
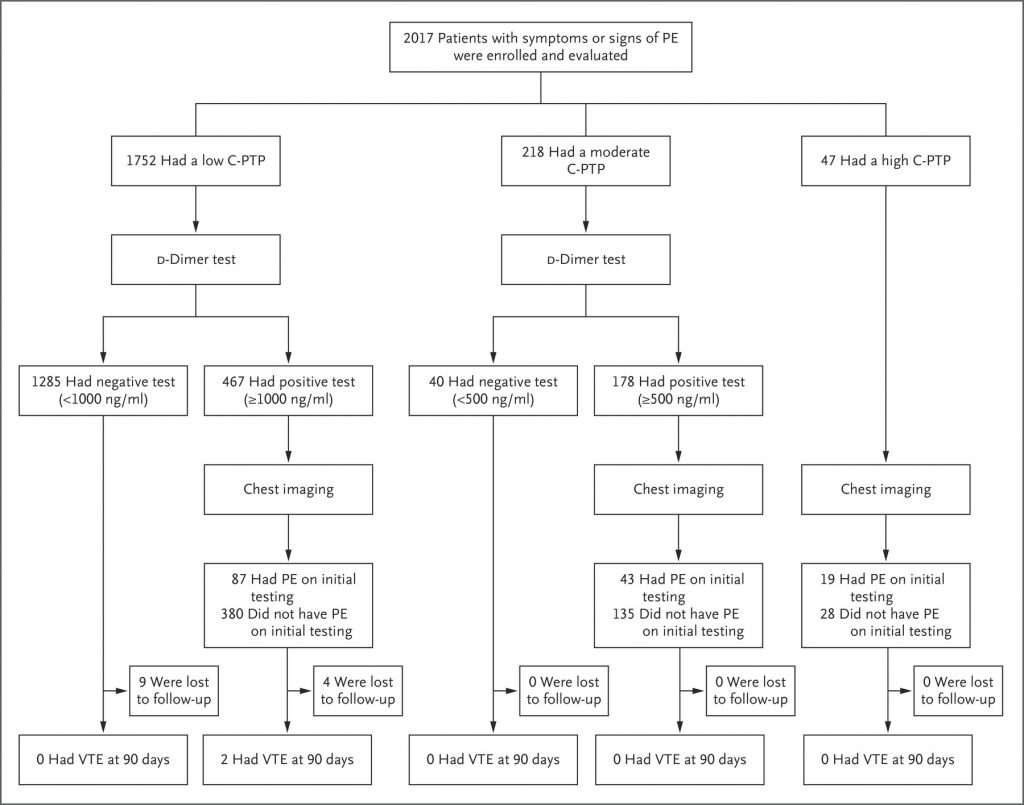
C Kearon et al. N Engl J Med 2019;381:2125-2134.
Secondary outcomes
Total of 7 major and 23 minor bleeding episodes were reported. There were 34 deaths during follow-up and none of them were attributed to pulmonary embolism. Most of the deaths were due to cancer. Most of the bleedings were gastrointestinal or genitourinary in nature.
When PEGeD strategy in all patients (all pretest probability subgroups combined) was compared to standard strategy (PE ruled out in low probability when D-dimer < 500 ng/ml), age-adjusted D-dimer (cut-off 50 x age for patients > 50 years of age) or YEARS strategy it resulted in less use of chest imaging.
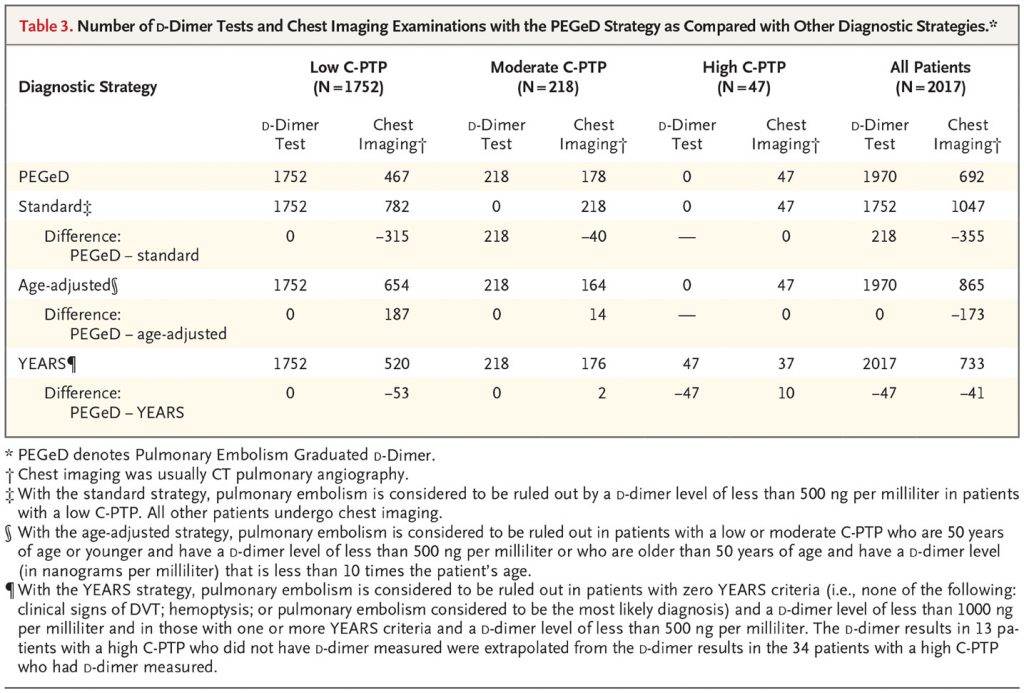
C Kearon et al. N Engl J Med 2019;381:2125-2134.
What are the results of this study?
See above.
How precise are the results?
Sub-analysis of all patients and patients with low pretest probability were precise. Patients with moderate or high pretest probability had low sample sizes and lower precision.
Do you believe the results?
Yes. Very few patients were lost to follow-up and hazard ratio for developing PE was higher in moderate and high probability groups.
Discussion
This study has shown that higher D-dimer cut-off of 1000 ng/ml can be used in outpatients with suspected pulmonary embolism and low pretest clinical probability. This has already been demonstrated in ADJUST-PE Study using age-adjusted D-dimer. As mentioned in the article, the main benefit of using less chest imaging is demonstrated in cohort of patients with low probability and D-dimer of 500 – 999 ng/ml. In total of 315 patients none of them developed VTE (95% CI 0% – 1.2%).
Results are consistent to other available evidence suggesting D-dimer can be safe to rule out PE in certain groups based on clinical pretest probability (BMJ 2010;340:c1475, Haematologica. 2012;97(10):1507-13, Thromb Haemost. 2012;107(1):167-71, BMJ. 2013;346:f2492).
Do the results of this study fit with other available evidence?
Yes.
In patients with moderate probability and negative D-dimer (<500 ng/ml) evidence is not that strong. Only 218 patients were analysed and only 40 of them had negative D-dimer. While in none of those patients with negative D-dimer developed VTE at 90 days, 95% is 0% – 8.76% and sample size is too small to make conclusions.
Prevalence of PE in this study was 7% (149 patients). In comparison in meta-analysis by Schouten et al. prevalence ranged from 5.1% to 39%. Negative predictive value of PEGeD can be lower in the general population. Prevalence of PE in suspected patients in Europe was significantly higher (26.5%) compared to US population (7.1%) as demonstrated in this study. Unfortunately it means that the results cannot be applied to local population.
Can the results be applied to local population?
No.
Amongst other drawbacks of the study, it is not clear how many patients were enrolled and how many patients were diagnosed with suspected PE in total in the participating centres, hence selection bias might be present. This is only an observational study, therefore randomised controlled trials should be run before this diagnostic algorithm can be safely used in practice and recommended in guidelines.
What are the implications of this study for practice?
Outpatients with low clinical pretest probability based on Wells’ score with negative D-dimer can have pulmonary embolism safely ruled out. Similar might be true for patients with moderate probability, although the sample size was small.
Author and attribution
Dr Timotej Vataha is a trainee physician in Internal Medicine Training programme in the UK. No conflicts of interest have been disclosed.
12 questions designed from Cohort study checklist by Critical Appraisal Skills Programme have been used under non-commercial CC 3.0 license.
References
Critical Appraisal Skills Programme (2018). CASP (Cohort Study) Checklist. [online] Available at: https://casp-uk.net/wp-content/uploads/2018/03/CASP-Cohort-Study-Checklist-2018_fillable_form.pdf. Accessed: December 20, 2019.
Nice.org.uk. (2019). Overview | Venous thromboembolic diseases: diagnosis, management and thrombophilia testing | Guidance | NICE. [online] Available at: https://www.nice.org.uk/Guidance/CG144 [Accessed 20 Dec. 2019].
Righini, M., Van Es, J., Den Exter, P., Roy, P., Verschuren, F., Ghuysen, A., Rutschmann, O., Sanchez, O., Jaffrelot, M., Trinh-Duc, A., Le Gall, C., Moustafa, F., Principe, A., Van Houten, A., Ten Wolde, M., Douma, R., Hazelaar, G., Erkens, P., Van Kralingen, K., Grootenboers, M., Durian, M., Cheung, Y., Meyer, G., Bounameaux, H., Huisman, M., Kamphuisen, P. and Le Gal, G. (2014). Age-Adjusted D-Dimer Cutoff Levels to Rule Out Pulmonary Embolism. JAMA, 311(11), p.1117.
Douma, R., le Gal, G., Sohne, M., Righini, M., Kamphuisen, P., Perrier, A., Kruip, M., Bounameaux, H., Buller, H. and Roy, P. (2019). Potential of an age adjusted D-dimer cut-off value to improve the exclusion of pulmonary embolism in older patients: a retrospective analysis of three large cohorts.
Douma, R., Tan, M., Schutgens, R., Bates, S., Perrier, A., Legnani, C., Biesma, D., Ginsberg, J., Bounameaux, H., Palareti, G., Carrier, M., Mol, G., Le Gal, G., Kamphuisen, P. and Righini, M. (2012). Using an age-dependent D-dimer cut-off value increases the number of older patients in whom deep vein thrombosis can be safely excluded. Haematologica, 97(10), pp.1507-1513.
Mos, I., Douma, R., Erkens, P., Durian, M., Nizet, T., van Houten, A., Hofstee, H., ten Cate, H., Ullmann, E., Büller, H., Huisman, M., Kamphuisen, P. and van Es, J. (2012). The combination of four different clinical decision rules and an age-adjusted D-dimer cut-off increases the number of patients in whom acute pulmonary embolism can safely be excluded. Thrombosis and Haemostasis, 107(01), pp.167-171.
Schouten, H., Geersing, G., Koek, H., Zuithoff, N., Janssen, K., Douma, R., van Delden, J., Moons, K. and Reitsma, J. (2013). Diagnostic accuracy of conventional or age adjusted D-dimer cut-off values in older patients with suspected venous thromboembolism: systematic review and meta-analysis. BMJ, 346(may03 1), pp.f2492-f2492.
Penaloza, A., Kline, J., Verschuren, F., Courtney, D., Zech, F., Derrien, B., Vielle, B., Armand-Perroux, A., Thys, F. and Roy, P. (2012). European and American suspected and confirmed pulmonary embolism populations: comparison and analysis. Journal of Thrombosis and Haemostasis, 10(3), pp.375-381.